Quality inspection of infusion bags presents challenges. It must meet strict requirements to guarantee product and patient safety. VITRONIC guarantees a cost-efficient yet precise inspection. With automation, you’ll achieve savings compared with manual inspection and enjoy improved results.
Smart Production A System for Complex Cases
Modern Inspection Methods for Greater Quality and Efficiency
So, what solutions are available? Here, we provide an overview of new methods and present one concrete example.
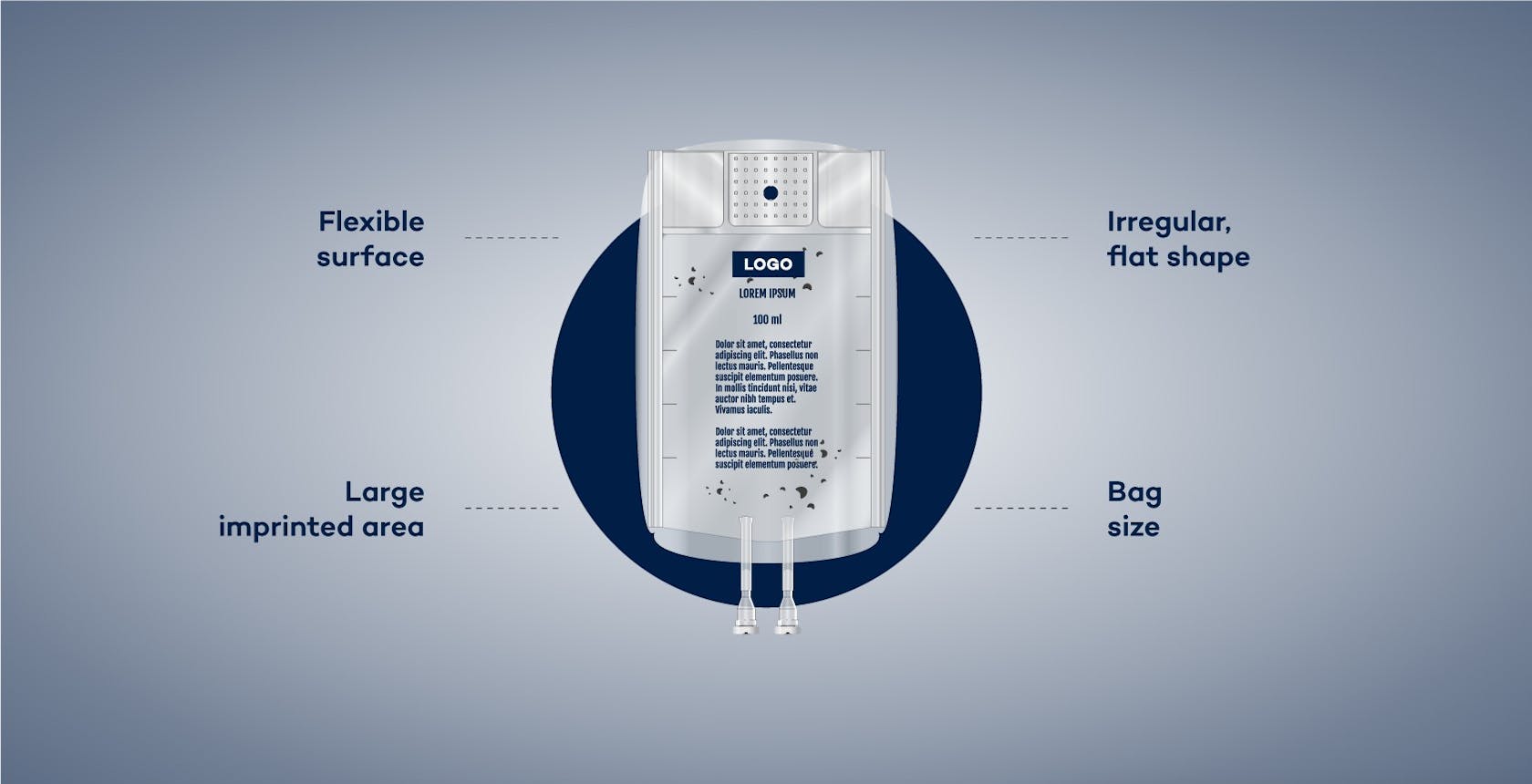
Products deemed “difficult to inspect” present a particular challenge for automatic visual inspection (AVI) in pharmaceutical filling and packaging processes. These products include infusion bags and bags used for parenteral nutrition, which hamper optical inspection due to their particular shape, size, and large printed areas.
Defect Detection Versus Reject Rates
Today, pharmaceutical manufacturers still complain that the results of inspections during production processes often fail to meet their expectations. Above all, they take issue with large volumes of defect-free products being scrapped and the failure to identify defects such as incorrect labeling or foreign particles. However, these two aspects are in conflict with one another. If inspection accuracy is increased, more defects will be detected.
On the other hand, this may also increase the number of false rejects, i.e., OK parts incorrectly classified as NOK parts. If, in contrast, inspection accuracy is low, it can be expected that fewer products will be scrapped. This then increases the possibility of some defects slipping through the net—in turn putting patient safety at risk.
Difficult to Inspect
In particular, the visual inspection of large-volume packaging bags, such as infusion bags or multi-chamber bags for parenteral nutrition, often produces unsatisfactory results. This is due to certain properties of these bags:
The irregular shape of the bags hinders the detection of printed text, codes, and logos, as these may become distorted by the curved surface. In addition, factors such as shadows, light reflections, and undefined air bubbles, make it difficult to distinguish between particles and acceptable influences.
The unique shape of the bags requires a test arrangement which allows a fast and robust inspection combined with high precision, this is possible in motionless condition only!
As large areas of the foils have usually been printed on, particles are not easily identified and are frequently interpreted incorrectly due to being partially covered by the printing. Finally, a challenge is presented by the fact that large-volume bags have an extensive area for inspection. The camera resolution for particle inspection is relatively low and therefore imprecise if the camera sensor is not adjusted.
A Multi-Stage Inspection Method
So, what solutions are available to the many and varied challenges that arise when inspecting large-format bags? With an effective illumination setup, careful positioning of the object to be inspected, and selection of the right background, it is easier to detect shadows and light reflections, as well as particle defects, print defects, and material defects.
To ensure a reliable detection of particles and an accurate distinction between particles and air bubbles or imprints, precise classification and fine-tuning is also required. For this purpose, the hardware and software in the inspection system must be perfectly aligned and adjusted to the specific requirements of the process environment.
Precise detection of the positioning of the printing on the bag is also essential in order for the software to reliably distinguish between this and particles on the bag. Finally, a high-resolution camera is the method of choice for capturing in detail the inspection area of the bags.
Automatic Visual Inspection at B. Braun
As a rule, it can be said that products classified as “difficult to inspect”, such as large-format bags, make many demands on automated optical inspection. A range of measures is required to overcome the individual challenges presented by the inspection tasks. One example of successful multi-stage inspection is the print inspection solution for multi-chamber bags that was developed by VITRONIC for B. Braun, a global leader in pharmaceutical and medical devices.
The optical inspection system was successfully implemented at the B. Braun headquarters in Melsungen at the end of last year. And the project managers are delighted with the results.
VITRONIC provided us with optimal support in the form of a new inspection solution. Since the start of the production process, we’ve had less than 1 percent scrap. That’s an excellent result.
Conclusion
In Brief
- Automatic inspection prevents large volumes of defect-free products from being scrapped
- A single static inspection ensures 100 percent accuracy
- With VITRONIC, you significantly reduce downtimes and enhance your productivity